Understanding Food-Grade Vs. Biocompatibility For Medical Device Materials | Medical Product Outsourcing
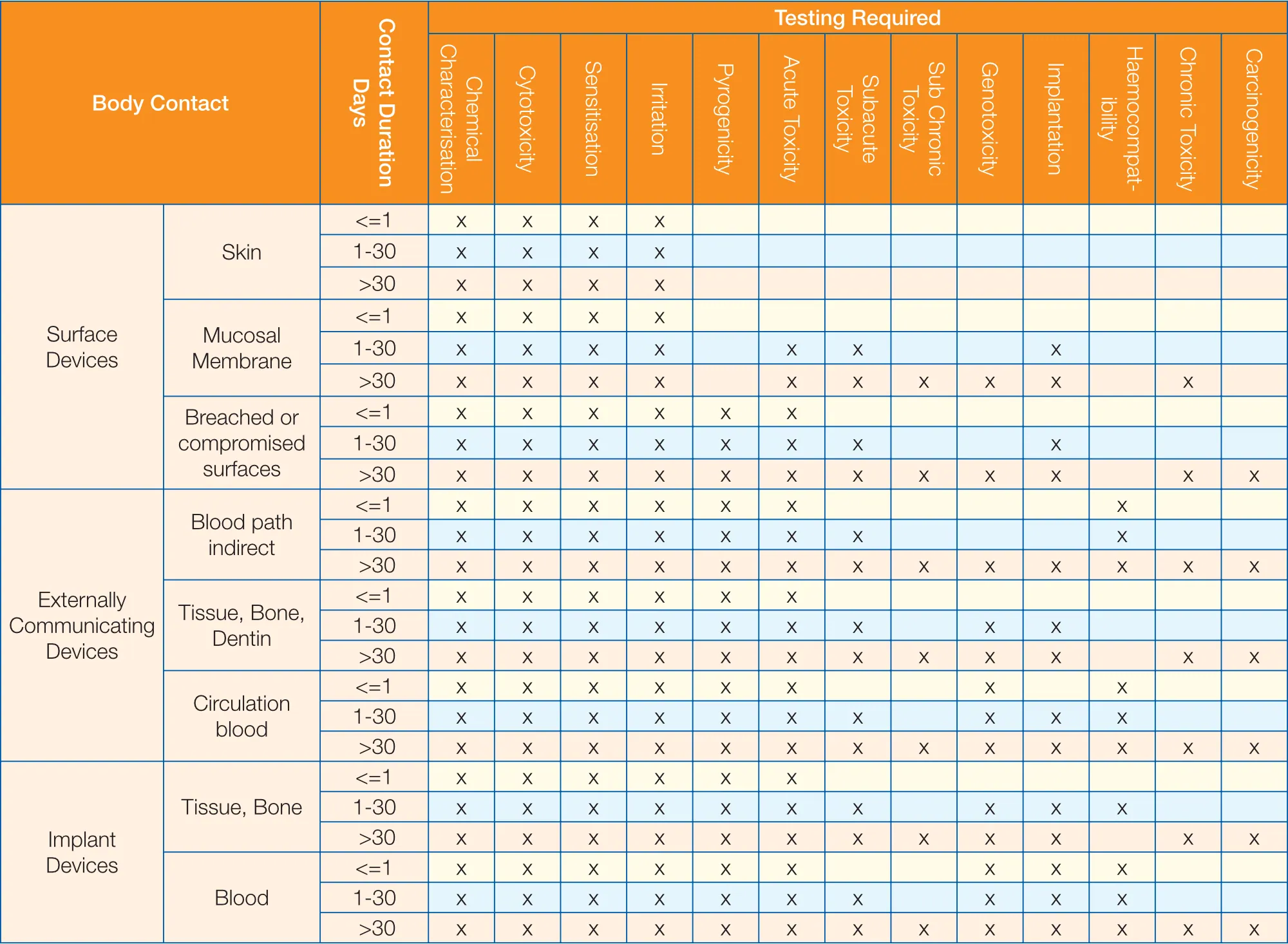
Biological Compatibility of Medical Service (ISO 10993) - Superlab CRO -health food, medical device, chemicals, pharmaceutical research (GLP&TAF accredited)

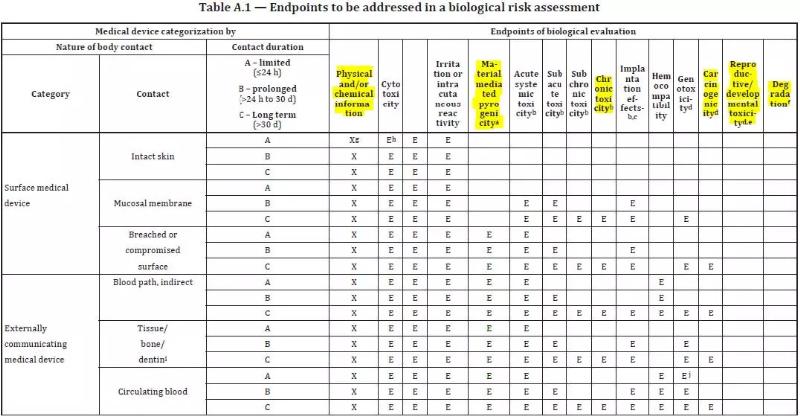
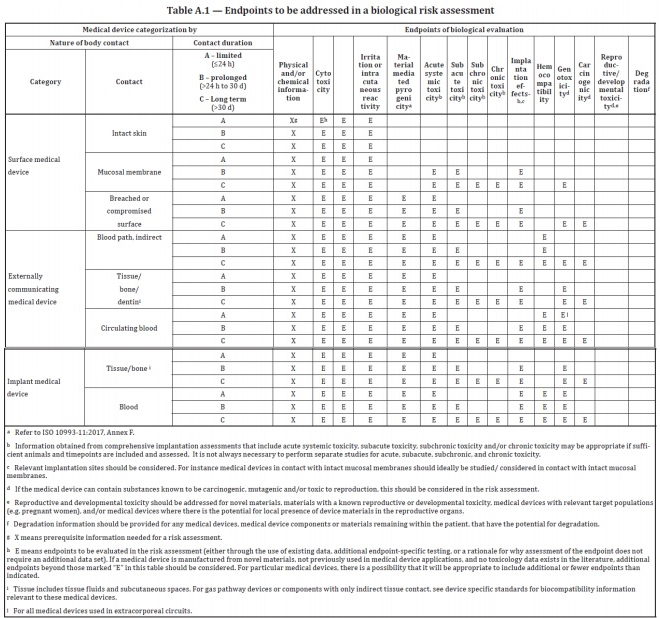
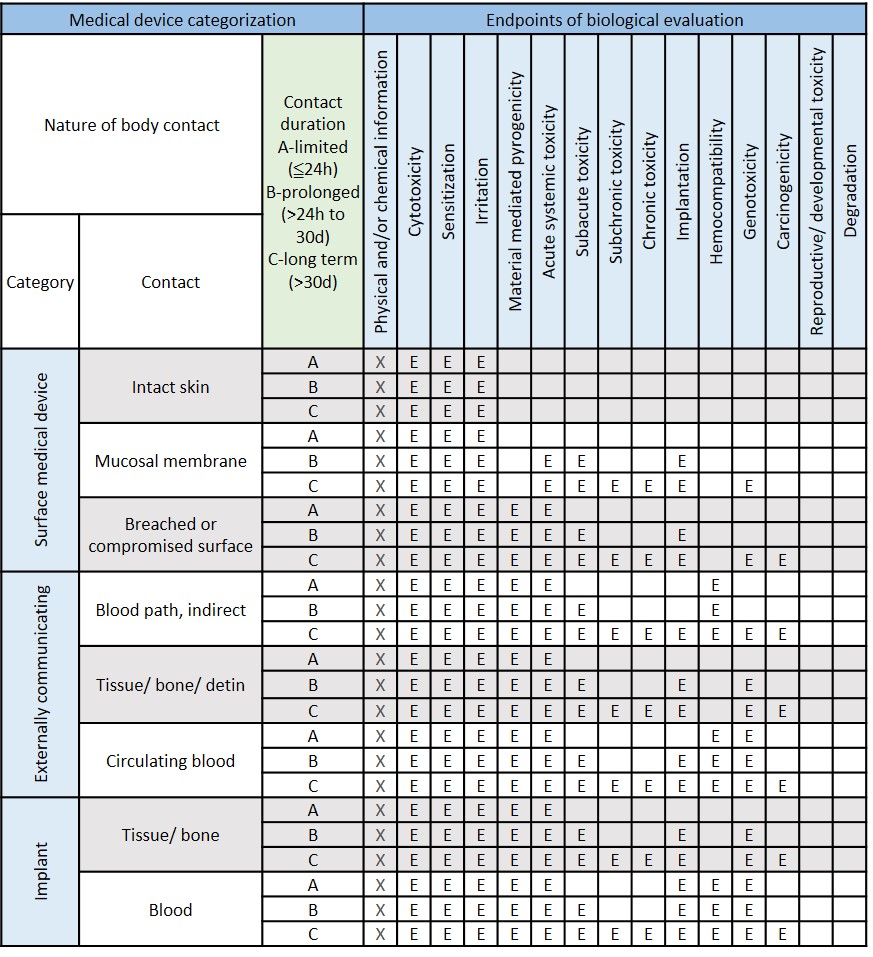
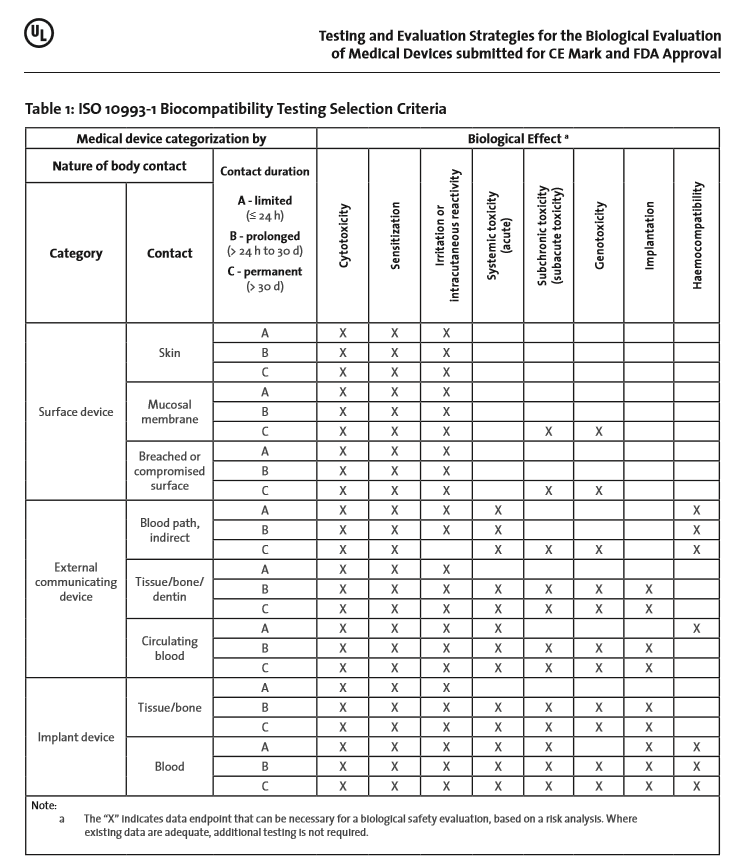
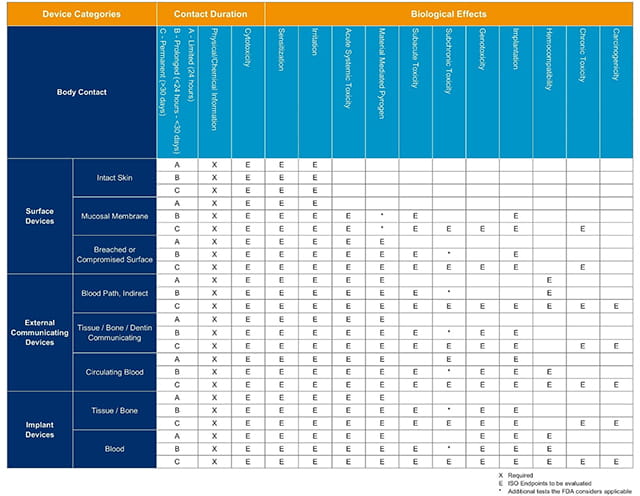
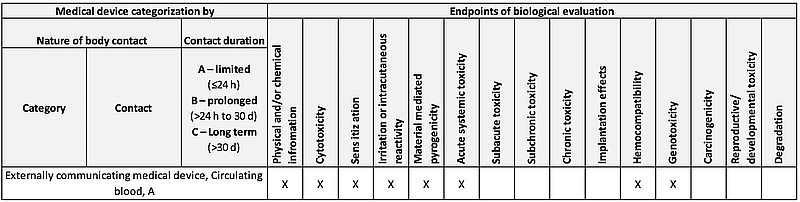
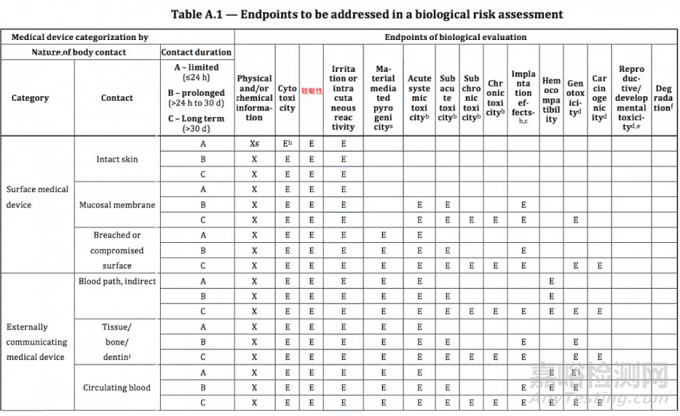
ISO 10993-1:2018(en), Biological evaluation of medical devices — Part 1: Evaluation and testing within a risk management process

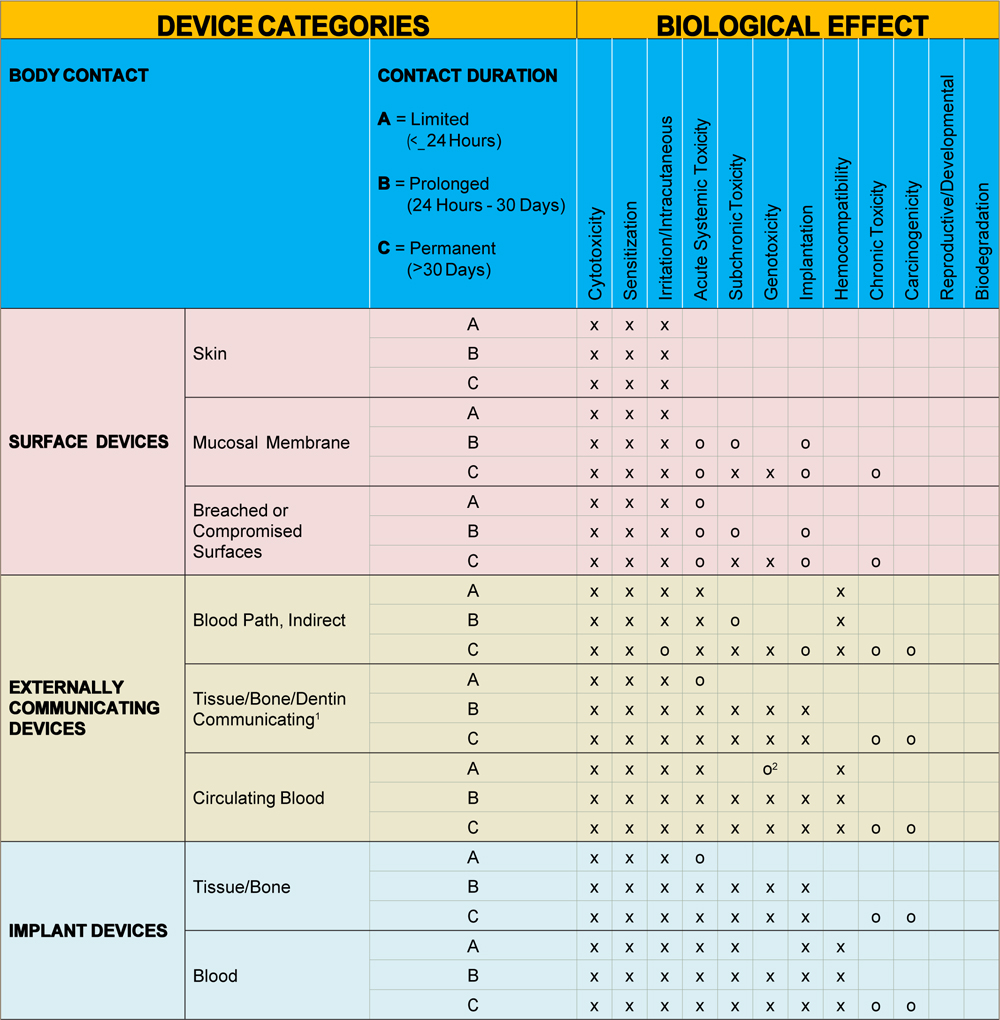
Towards a Logic-Based Extension of a Relational Software Tool for Coherent Technical Documentation of Medical Devices - In Compliance Magazine